The TRC is focused on the recruitment of patients into early stage proof-of-concept trials. This is different from trials run by many contract research organizations, which typically recruit healthy human volunteers.
Early treatment of patients with a potential therapeutic provides evidence for efficacy in the target treatment population. It also allows measurements of key biochemical, genetic, and radiological parameters to be made in affected individuals.
Open to Collaboration
We are conducting a large number (>200) of active protocols, approximately 35 percent of which are industry-sponsored studies.
We have also introduced a new partnership model in which there is a single administrative docking point to streamline contract negotiations and IRB approvals to better meet the timelines typical of industry studies.
For smaller biotech companies with limited in-house clinical expertise, the TRC can bring disease experts from Mass General and beyond to assist in building clinical trial protocols that ask and answer key questions in the evaluation of a novel therapeutic.
About Our Center
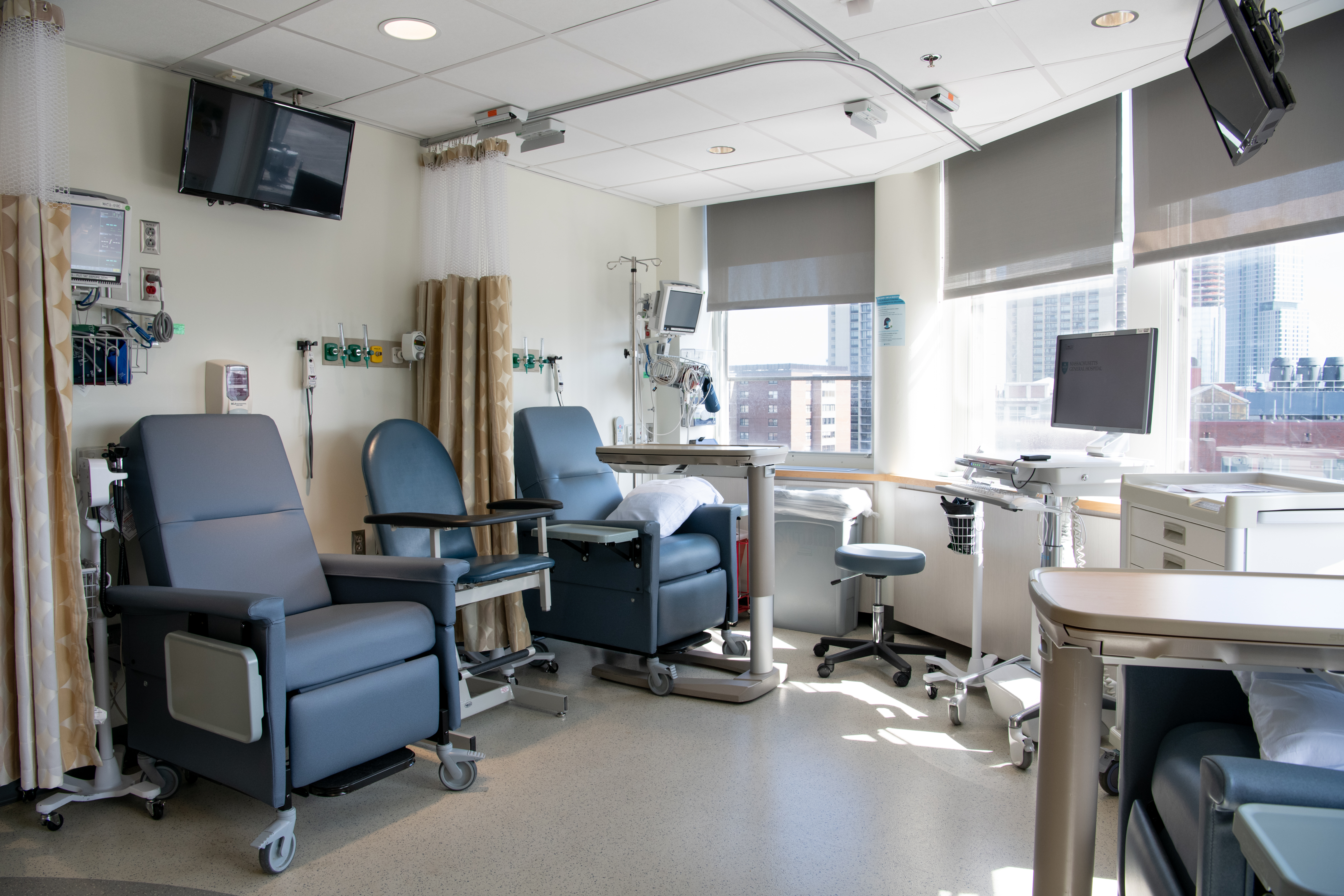
The TRC offers all the advantages of an inpatient hospital care in a setting that is specialized for clinical research.
The center includes up to 20 inpatient beds and four infusion chairs. Each bed has central monitoring and oxygen at the headwall. There are two private rooms with air flow monitoring suitable for studies involving gene therapy vectors or other agents where containment of biological materials is critical.
The TRC staff includes expert research nurses, nurse practitioners, research nutritionists, laboratory technicians, and project managers. Both adult and pediatric study participants can be accommodated.
The laboratory and imaging technical capabilities of the hospital (e.g., genomics, specialized immunological assays, PET scanning) are available to illuminate key research questions.
The TRC can draw from a pool of 3 million patients cared for in the Mass General/Partners HealthCare system and match them with the right clinical investigator from the 2,500 physician scientists at Mass General, who are leading experts in their respective disciplines.